 Thus the number of electrons employed in double bonds = 8ĭeducting that from the total valence electrons we get 10 electrons remaining. There are 2 oxygen atoms in the compound, thus = 6*2 = 12Īfter drawing the skeletal structure, we can see that none of the atoms can achieve their octet with single bonds. In SO2, the sulfur’s valence electron = 6 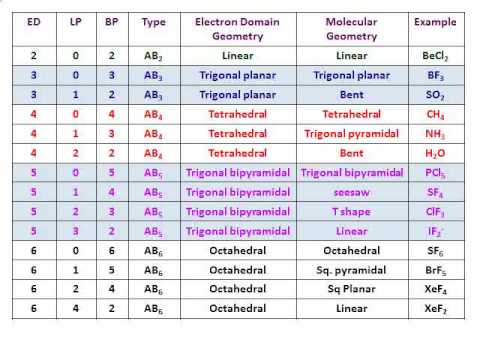
Now let’s see the lewis structure of SO2. The total number of bonding electrons around the boron is 8 (full octet). The number of non-bonded electrons is zero. The number of valence electrons for boron is 3. This formula explicitly indicates the relationship between the number of bonding electrons and their link to how many are formally “ kept ” by the atom.įor example, applying this to BH4 we get: Step 6 – At last, it’s important to analyze if all the atoms are having their lowest possible formal charge.įormal charge calculation can be done using:-įormal charge = –.Step 5 – Giving double or triple bonds is fundamental if it is needed for fulfilling the octet rule for all atoms.Always start with the electronegative atoms then bring them to the electropositive ones. Step 4 – Next, our work is achieving the octet of the atoms with the resting electrons, after the formation of the single bonds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
